Antibiotic treatments have detrimental effects on the microbiome and lead to antibiotic resistance. To develop a phage therapy against a diverse range of clinically relevant Escherichia coli, we screened a library of 162 wild-type (WT) phages, identifying eight phages with broad coverage of E. coli, complementary binding to bacterial surface receptors, and the capability to stably carry inserted cargo. Selected phages were engineered with tail fibers and CRISPR–Cas machinery to specifically target E. coli. We show that engineered phages target bacteria in biofilms, reduce the emergence of phage-tolerant E. coli and out-compete their ancestral WT phages in coculture experiments. A combination of the four most complementary bacteriophages, called SNIPR001, is well tolerated in both mouse models and minipigs and reduces E. coli load in the mouse gut better than its constituent components separately. SNIPR001 is in clinical development to selectively kill E. coli, which may cause fatal infections in hematological cancer patients. Phage engineered with tail fibers and CRISPR–Cas reduce Escherichia coli load in animals.
Research and publish the best content.
Get Started for FREE
Sign up with Facebook Sign up with X
I don't have a Facebook or a X account
Already have an account: Login
 Your new post is loading... Your new post is loading...
 Your new post is loading... Your new post is loading...
|




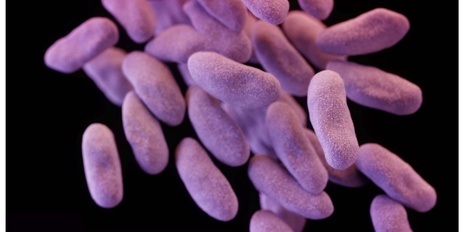






The emergence of antibiotic-resistant pathogens represents a major global challenge. In numerous disease areas, current medical practice relies on increasingly aggressive therapies, which can sometimes result in patients experiencing life-threatening infections, including those caused by antibiotic-resistant bacteria. SNIPR001's mechanism of action, as a CRISPR-armed phage therapeutic that specifically targets and eradicates E. coli in the gut, is designed to prevent infections from spreading into the bloodstream and represents a promising advancement against antibiotic-resistant pathogens. This publication represents a significant validation of the ground-breaking technology research done at the laboratories and collaborators of SNIPR Biome, the company pioneering CRISPR-based microbial gene therapy, and also opens up the possibility of targeting other pathogens. SNIPR001 has been granted a Fast-Track designation by the US Food and Drug Administration, was supported by CARB-X, and SNIPR is currently conducting a Phase 1 trial in the US to evaluate its safety and efficacy in reducing E. coli in the gut without disturbing the overall gut microbiome.