Research and publish the best content.
Get Started for FREE
Sign up with Facebook Sign up with X
I don't have a Facebook or a X account
Already have an account: Login
 Your new post is loading... Your new post is loading...
 Your new post is loading... Your new post is loading...
|
|






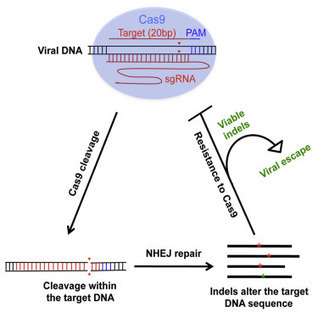







A new study from Tel Aviv University proposes a new and unique AIDS treatment that could be developed into a vaccine or a one-time treatment for HIV patients. The study examined the engineering of B-type white blood cells in the patient's body to secrete anti-HIV antibodies in response to the virus. The technique developed in his laboratory uses B-white blood cells that would be genetically modified inside the patient's body to secrete neutralizing antibodies against the HIV virus. The gene editing was done with a CRISPR system. The researchers are able to engineer the B cells inside the patient's body using two viral vectors from the AAV family, one encodes the desired antibody and the second encodes the CRISPR system. When CRISPR cuts the desired site in the genome of the B cells it directs the introduction of the desired gene: the gene coding for the antibody against the HIV virus. On the basis of this study, we can hope that in the next few years we will be able to produce a drug against AIDS in this way, but also against other infectious diseases, for certain types of cancer caused by a virus, such as cervical cancer.