Editing a single nucleotide in the largest human gene is sufficient to restore dystrophin production and myocyte function in Duchenne muscular dystrophy. The new research uses base editing and prime editing that only nicks a single strand of DNA and reduces the risk of damaging genetic changes.
Research and publish the best content.
Get Started for FREE
Sign up with Facebook Sign up with X
I don't have a Facebook or a X account
Already have an account: Login
 Your new post is loading... Your new post is loading...
 Your new post is loading... Your new post is loading...
|
|






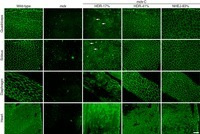





To restore dystrophin production and myocyte function in patients with Duchenne muscular dystrophy (DMD), several CRISPR strategies have been tried to correct some of the hundreds of thousands of documented mutations in the dystrophin gene (DMD). Among these strategies, researchers at the University of Texas Southwestern Medical Center in Dallas, Texas, have developed a novel CRISPR gene editing strategy for DMD therapy. This was tested in animal and human DMD models where the researchers deleted exon 51 in the DMD gene which disrupted the dystrophin reading frame and generated a premature stop codon in exon 52. The researchers were able to restore the reading frame with both basic and main editing by introducing exon skipping and reframing, respectively, through their "single-swap" ABE strategy, i.e., through editing a single letter in the splice acceptor or donor site can cause exon skipping. The researchers targeted the splice acceptor or splice donor site of exon 50 or 52 and the single nucleotide modification restored dystrophin expression in human cardiomyocytes and contractile function was normalized using master editing. However, one challenge is the high doses required to deliver expression throughout the body.