Your new post is loading...
 Your new post is loading...
This topic is devoted to Neuro-immunology, covering various neurological disorders with confirmed or possible immunological mechanisms. This year (2022), many students are choosing Neurology and neurological diseases for their Scoop.it exercise. It will stimulate me for improving this topic (tagging...) to help them navigate the curated resources. Some key points: We developed original research on Choroid Plexuses in ageing and Alzheimer's disease. https://www.scoop.it/topic/neuroimmunology?q=choroid+plexuses The topic of autoimmunity at large, became very fashionable recently. https://www.scoop.it/topic/neuroimmunology?q=autoimmunity with prescription of many biological tests for diagnosis of difficult neurological clinical disorders. We were also very much involved, 10 years ago in the development of CSFTCs detection, enumeration and analysis in carcinoma meningitis, a rare but dramatic issue in breast, lung ... cancers and melanomas. Papers on https://www.scoop.it/topic/nancytomique
Scientists identify the brain cells that regulate inflammation, and pinpoint how they keep tabs on the immune response.
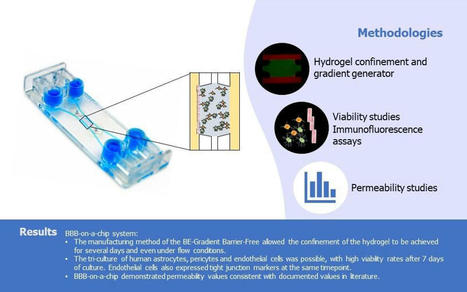
🔬 Exciting Developments in Blood-Brain Barrier Modeling: Introducing the BE-Gradient Barrier-Free Device 🔬
We are thrilled to share a landmark publication…
Adequate drug delivery across the blood-brain barrier is a critical factor in treating central nervous system (CNS) disorders. Inspired by swimming fish and the microstructure of the nasal cavity, Prof.
Studies of the choroid plexus lag behind those of the more widely known blood-brain barrier, despite a much longer history. This review has two overall aims. The first is to outline long-standing areas of research where there are unanswered questions, such as control of cerebrospinal fluid (CSF) sec …
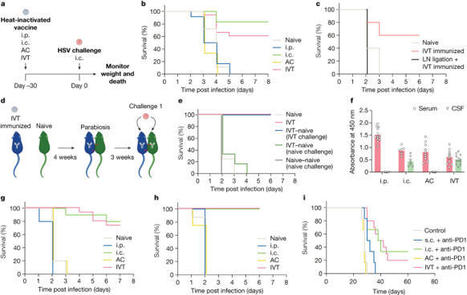
The eye, an anatomical extension of the central nervous system (CNS), exhibits many molecular and cellular parallels to the brain. Emerging research demonstrates that changes in the brain are often reflected in the eye, particularly in the retina1. Still, the possibility of an immunological nexus between the posterior eye and the rest of the CNS tissues remains unexplored. Here, studying immune responses to herpes simplex virus in the brain, we observed that intravitreal immunization protects mice against intracranial viral challenge. This protection extended to bacteria and even tumours, allowing therapeutic immune responses against glioblastoma through intravitreal immunization. We further show that the anterior and posterior compartments of the eye have distinct lymphatic drainage systems, with the latter draining to the deep cervical lymph nodes through lymphatic vasculature in the optic nerve sheath. This posterior lymphatic drainage, like that of meningeal lymphatics, could be modulated by the lymphatic stimulator VEGFC. Conversely, we show that inhibition of lymphatic signalling on the optic nerve could overcome a major limitation in gene therapy by diminishing the immune response to adeno-associated virus and ensuring continued efficacy after multiple doses. These results reveal a shared lymphatic circuit able to mount a unified immune response between the posterior eye and the brain, highlighting an understudied immunological feature of the eye and opening up the potential for new therapeutic strategies in ocular and CNS diseases. A study provides evidence for a shared lymphatic circuit that connects the posterior eye and the brain, allowing the generation of immune responses to protect the CNS against pathogens and tumours following intravitreal immunization.
/PRNewswire/ -- Oscotec Inc. and ADEL Inc. announced the dosing of the first healthy participant in its first-in-human study of ADEL-Y01 for the treatment of...
In the 3D Brain Atlas you can see many of the brain’s structures from a 360 degree viewpoint, make various sagittal incisions through the brain, read more about the structures, and download images of the brain that you can use in your own presentations.
The arachnoid barrier delineates the border between the central nervous system and dura mater. Although the arachnoid barrier creates a partition, communication between the central nervous system and the dura mater is crucial for waste clearance and immune surveillance1,2. How the arachnoid barrier balances separation and communication is poorly understood. Here, using transcriptomic data, we developed transgenic mice to examine specific anatomical structures that function as routes across the arachnoid barrier. Bridging veins create discontinuities where they cross the arachnoid barrier, forming structures that we termed arachnoid cuff exit (ACE) points. The openings that ACE points create allow the exchange of fluids and molecules between the subarachnoid space and the dura, enabling the drainage of cerebrospinal fluid and limited entry of molecules from the dura to the subarachnoid space. In healthy human volunteers, magnetic resonance imaging tracers transit along bridging veins in a similar manner to access the subarachnoid space. Notably, in neuroinflammatory conditions such as experimental autoimmune encephalomyelitis, ACE points also enable cellular trafficking, representing a route for immune cells to directly enter the subarachnoid space from the dura mater. Collectively, our results indicate that ACE points are a critical part of the anatomy of neuroimmune communication in both mice and humans that link the central nervous system with the dura and its immunological diversity and waste clearance systems. Arachnoid cuff exit points create openings in the arachnoid barrier enabling the drainage of cerebrospinal fluid and exchange of molecules and cells between the dura and the subarachnoid space, therefore physically connecting the brain and the dura.
Microglia are thought to drive neuroinflammation in age-related macular degeneration (AMD). Here, Yu et al. identified a protective microglia population that mi
Cerebrospinal fluid (CSF) circulates around the brain and the spinal column, providing mechanical support and nourishment, and helping to clea
We profiled human central nervous system (CNS)-associated macrophages (CAMs) in anatomically dissected CNS interface tissue from typical, fetal and glioblastoma-affected brains using single-cell multi-omics and spatially resolved transcriptomic techniques.
|
Great initiative from Fluigent on organ-on-chip technologies! Lot of invited speakers and the FLUIGENT team ready to show you some cool equipment and…
Sign in or join now to see posts like this one and more.
Impaired lymphatic drainage of brain waste occurs in aging and disease, but the cause remains elusive. Here, Rustenhoven et al. describe how meningeal immune ce
Hopes are high that engineered immune cells, which are already in use to treat blood cancer, will halt the progression of a degenerative autoimmune disorder.
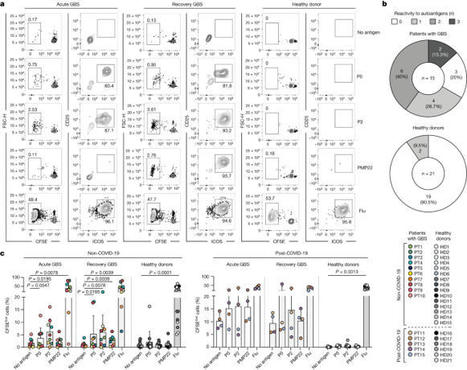
Guillain–Barré syndrome (GBS) is a rare heterogenous disorder of the peripheral nervous system, which is usually triggered by a preceding infection, and causes a potentially life-threatening progressive muscle weakness1. Although GBS is considered an autoimmune disease, the mechanisms that underlie its distinct clinical subtypes remain largely unknown. Here, by combining in vitro T cell screening, single-cell RNA sequencing and T cell receptor (TCR) sequencing, we identify autoreactive memory CD4+ cells, that show a cytotoxic T helper 1 (TH1)-like phenotype, and rare CD8+ T cells that target myelin antigens of the peripheral nerves in patients with the demyelinating disease variant. We characterized more than 1,000 autoreactive single T cell clones, which revealed a polyclonal TCR repertoire, short CDR3β lengths, preferential HLA-DR restrictions and recognition of immunodominant epitopes. We found that autoreactive TCRβ clonotypes were expanded in the blood of the same patient at distinct disease stages and, notably, that they were shared in the blood and the cerebrospinal fluid across different patients with GBS, but not in control individuals. Finally, we identified myelin-reactive T cells in the nerve biopsy from one patient, which indicates that these cells contribute directly to disease pathophysiology. Collectively, our data provide clear evidence of autoreactive T cell immunity in a subset of patients with GBS, and open new perspectives in the field of inflammatory peripheral neuropathies, with potential impact for biomedical applications. Autoreactive T cells that target myelin antigens in the peripheral nerves are present in patients with the demyelinating form of Guillain–Barré syndrome, and these T cells are likely to contribute to disease pathophysiology.
Researchers discovered a crucial network of lymphatic vessels at the back of the nose, which significantly contributes to draining cerebrospinal fluid (CSF) from the brain.
Min et al. show mural cells physically contact macrophages in the dura to regulate trafficking of CNS antigen-specific T cells. Mural cells are altered in presy
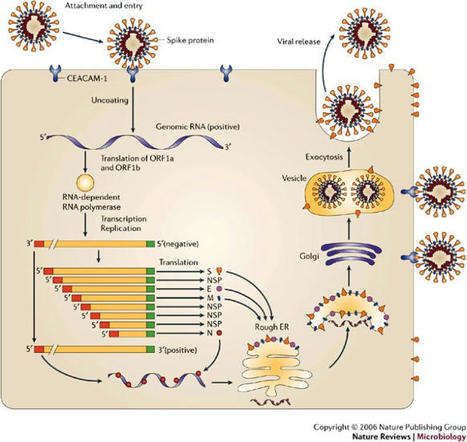
The need for a balance between pathogen elimination and protection from cellular damage means that the central nervous system (CNS) is a partially protected niche that some pathogens can exploit. Here, the authors discuss the immune regulation of acute and persistent CNS infection by coronaviruses, using mouse hepatitis virus as a model. Several viruses infect the mammalian central nervous system (CNS), some with devastating consequences, others resulting in chronic or persistent infections associated with little or no overt pathology. Coronavirus infection of the murine CNS illustrates the contributions of both the innate immune response and specific host effector mechanisms that control virus replication in distinct CNS cell types. Despite T-cell-mediated control of acute virus infection, host regulatory mechanisms, probably designed to protect CNS integrity, contribute to the failure to eliminate virus. Distinct from cytolytic effector mechanisms expressed during acute infection, non-lytic humoral immunity prevails in suppressing infectious virus during persistence.
|
 Your new post is loading...
Your new post is loading...
 Your new post is loading...
Your new post is loading...