Your new post is loading...

|
Scooped by
Juan Lama
|
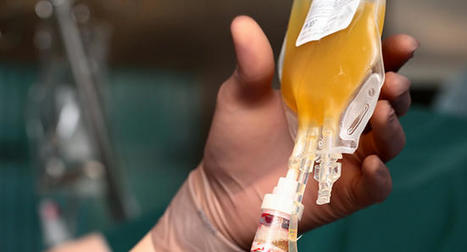
Jan. 15, 2021 -- Blood transfusions from recovered COVID-19 patients that contained high antibody levels reduced the number of COVID-19 deaths among other hospitalized patients, according to a new study by Mayo Clinic. If given early to patients with mild COVID-19, the treatments slowed the progression of disease, they wrote in the New England Journal of Medicine. “I think it behooves the medical community to continue to innovate and test therapies for treatment. Realistically, we’re months away from having a substantial number of people vaccinated,” R. Scott Wright, MD, coordinator of the Mayo Clinic’s COVID-19 plasma therapy program, told the Minneapolis Star-Tribune. Early in the pandemic, Mayo Clinic launched a nationwide program that linked hospitalized COVID-19 patients with donor plasma. The research team analyzed records for more than 3,000 patients to determine whether those who received blood with high levels of antibodies fared better than those who received blood with low antibody levels. Overall, patients who received antibody-rich plasma had a 25% lower chance of dying within 30 days. In addition, patients who received plasma within three days of their COVID-19 diagnosis had a lower risk of death than those who received transfusions later. Health outcomes were better for those who received plasma and didn’t need to be placed on a ventilator. “Patients who were on ventilator did not see a benefit,” Wright told the newspaper. “It was too late.” Hospitals should conserve their limited plasma supplies for patients who are most likely to benefit from a transfusion, Louis Katz, MD, the acting chief medical director of the Mississippi Valley Regional Blood Center in Iowa, wrote in an accompanying editorial in the New England Journal of Medicine. “Uncontrolled compassionate use of convalescent plasma…should be discouraged, even though clinicians recognize how difficult it can be to ‘just stand there’ at the bedside of a patient in the ICU,” he wrote. At his center, Katz said, about a fifth of plasma donations contain high enough antibodies to provide the benefits seen in the Mayo Clinic study. “Constraints on therapies for COVID-19 that are effective for limited patient populations are a powerful argument for continued consistent adherence to recommended nonpharmaceutical interventions and the rapid deployment and uptake of effective vaccines,” he wrote. Original Findings Publishes in NEJM (Jan. 13, 2020): https://doi.org/10.1056/NEJMoa2031893

|
Scooped by
Juan Lama
|
Laboratory diagnosis of SARS-CoV-2 infection (the cause of COVID-19) uses PCR to detect viral RNA (vRNA) in respiratory samples. SARS-CoV-2 RNA has also been detected in other sample types, but there is limited understanding of the clinical or laboratory significance of its detection in blood. Methods: We undertook a systematic literature review to assimilate the evidence for the frequency of vRNA in blood, and to identify associated clinical characteristics. We performed RT-PCR in serum samples from a UK clinical cohort of acute and convalescent COVID-19 cases (n=212), together with convalescent plasma samples collected by NHS Blood and Transplant (NHSBT) (n=212 additional samples). To determine whether PCR-positive blood samples could pose an infection risk, we attempted virus isolation from a subset of RNA-positive samples. We identified 28 relevant studies, reporting SARS-CoV-2 RNA in 0-76% of blood samples; pooled estimate 10% (95%CI 5-18%). Among serum samples from our clinical cohort, 27/212 (12.7%) had SARS-CoV-2 RNA detected by RT-PCR. RNA detection occurred in samples up to day 20 post symptom onset, and was associated with more severe disease (multivariable odds ratio 7.5). Across clinical and convalescent samples collected ≥28 days post symptom onset, 0/244 (0%, 95%CI 0.0-1.5%) had vRNA detected. Among our PCR-positive samples, cycle threshold (ct) values were high (range 33.5-44.8), suggesting low vRNA copy numbers. PCR-positive sera inoculated into cell culture did not produce any cytopathic effect or yield an increase in detectable SARS-CoV-2 RNA. Conclusions: vRNA was detectable at low viral loads in a minority of serum samples collected in acute infection, but was not associated with infectious SARS-CoV-2 (within the limitations of the assays used). This work helps to inform biosafety precautions for handling blood products from patients with current or previous COVID-19.

|
Scooped by
Juan Lama
|
New York Blood Center is seeking convalescent plasma from COVID-19 recovered patients who have been confirmed positive and free of symptoms for at least 14 days. NEW YORK – New York Blood Center (NYBC) will be the first blood center to collect blood plasma donations from people who have recovered from COVID-19 to treat other patients with advanced illness. In the treatment, known as convalescent plasma, the patient is transfused with the donor’s plasma with the goal of using the donor’s antibodies to help clear the virus more rapidly. Yesterday, the U.S. Food and Drug Administration (FDA) approved the treatment for use on a case by case basis and NYBC will be the first to collect plasma from recovered COVID-19 patients for this purpose. “We’re proud to partner with leading medical institutions from New York and beyond in developing this potential treatment,” said Beth H. Shaz, MD, Chief Medical and Scientific Officer at New York Blood Center. “If this treatment proves to be effective, we are prepared to quickly scale our process and activate our network to serve hospitals nationwide.” NYBC will collect the donations, process the plasma for infusion, and maintain a bank for hospitals to treat patients with serious or immediately life-threatening COVID-19 infections. Qualified donors will be referred to NYBC by area hospitals and the Food and Drug Administration (FDA) will approve patients for the treatment on a case-by-case basis. NYBC is one of the largest independent blood centers in the world. Its network serves local communities in New York, New Jersey, Connecticut, Pennsylvania, Delaware, Maryland, Virginia, Missouri, Kansas, Minnesota, Nebraska and Rhode Island. NYBC is prepared to expand convalescent plasma collections across these locations if it proves to be successful.

|
Scooped by
Juan Lama
|
Takeda announced that it has commenced the development of a plasma-derived therapy for COVID-19, a disease caused by severe SARS-CoV-2. The company will inform the members of the US Congress regarding the development of an anti-SARS-CoV-2 polyclonal hyperimmune globulin (H-IG) for the treatment of high-risk individuals with COVID-19. Takeda will also evaluate whether the firm’s currently marketed and pipeline products serve as effective treatments for infected patients. According to the company, the hyperimmune globulins are plasma derived-therapies that were earlier demonstrated as an effective treatment for severe acute viral respiratory infections and may serve as an option for COVID-19. The company is specialised in the research, development and manufacturing of a potential anti-SARS-CoV-2 polyclonal H-IG, which is referring to as TAK-888. Takeda’s vaccine business unit president and company’s COVID-19 response team co-lead Dr Rajeev Venkayya said: “We have identified relevant assets and capabilities across the company and are hopeful that we can expand the treatment options for patients with COVID-19 and the providers caring for them.” Currently, the Japanese pharmaceutical firm is in negotiations with multiple national health and regulatory agencies and health care partners in the US, Asia, and Europe to rapidly advance the research of TAK-888. Initially, the company plans to produce the therapy in an isolated within its manufacturing facility in Georgia to avoid a negative impact on its other plasma-derived therapies.
|

|
Scooped by
Juan Lama
|
Jill Horowitz stood outside the Quaker Ridge Shopping Center in New Rochelle, N.Y.—an early COVID-19 hotspot—in March, stopping shoppers as they walked into the grocery store. She handed them blue pamphlets soliciting volunteers for a Rockefeller University antibody research study. “I would say, ‘Would you like to help us find a cure?’” says Horowitz, executive director of strategic operations at Rockefeller’s Laboratory of Molecular Immunology. “I didn’t even have to mention coronavirus. This neighborhood was completely subsumed.” Within weeks—and after receiving more than 2,000 phone calls from volunteers—the university had selected more than 100 participants who had recovered from COVID-19 or had come into contact with someone who had the disease, says Michel Nussenzweig, head of the laboratory. From participants’ blood samples, he and his team isolated more than a dozen potent antibodies that “neutralized,” or deactivated, SARS-CoV-2, the virus that causes COVID-19, in a lab dish. The study is one of a growing number showing the body produces antibodies against this deadly disease. The findings suggest that therapies based on these proteins could be a promising approach. But experts caution that such therapies must clear several hurdles before they can be deployed against COVID-19. Our body naturally produces antibodies to help us fight infections. Many researchers believe that by isolating antibodies from people who have recovered from COVID-19 and then artificially reproducing them, we can develop therapies that could minimize symptoms and speed recovery from the disease. Some of the same scientists are also eyeing the prophylactic use of copied antibodies to stave off an infection in those who have not contracted the new coronavirus. (Therapies based on these so-called monoclonal antibodies are different from convalescent plasma treatments, which have also made headlines recently. In the latter, plasma is taken from people who have recovered from COVID-19 and transfused directly into those who are infected. The jury is still out on whether convalescent plasma is truly effective against the disease.)...

|
Scooped by
Juan Lama
|
The most comprehensive national study to date has found that convalescent plasma appears to be safe to use on COVID-19 patients, a promising development in the race to find a treatment for the deadly virus. But the study didn't determine whether the treatment works. A team of more than 5,000 doctors from over 2,000 hospitals and laboratories have been testing the experimental therapy, which involves transfusing the antibody-rich blood serum of recovered COVID-19 patients into people who are battling the illness. Of the 5,000 seriously ill patients who received blood plasma transfusions for the study, fewer than 1 percent experienced serious adverse events. The mortality rate seven days after treatment was 14.9 percent, but the researchers noted the infusion patients were already gravely ill and the rate "does not appear excessive." “We're very encouraged that the treatment is safe,” Dr. Michael Joyner, a physiologist at the Mayo Clinic and the principal investigator of the study, told NBC News. “That was really the first hurdle for us. And showing that it's safe gives us confidence to move on and begin to try to understand the efficacy.” The study, which was co-authored by researchers at several institutions including Michigan State University and Johns Hopkins, hasn’t undergone peer review or publication in a scientific journal. The results were posted on a public server, called MedRxiv, so that other scientists and physicians could quickly review the data.... The study notes that the most common complication of convalescent plasma therapy is transfusion associated circulatory overload (or TACO), which occurs when a patient cannot tolerate the increased blood volume that results from the transfusion of plasma. Researchers were also concerned about the possibility that the antibodies infused into patients could lead to the formation of new infections. Preprint of the Study available in medRxiv (May 14, 2020): https://doi.org/10.1101/2020.05.12.20099879

|
Scooped by
Juan Lama
|
Hoping to stem the toll of the state’s surging coronavirus outbreak, New York health officials plan to begin collecting plasma from people who have recovered and injecting the antibody-rich fluid into patients still fighting the virus. Gov. Andrew Cuomo announced the plans during a news briefing Monday. The treatment, known as convalescent plasma, dates back centuries and was used during the flu epidemic of 1918 — in an era before modern vaccines and antiviral drugs. Some experts say the treatment, although somewhat primitive, might be the best hope for combating the coronavirus until more sophisticated therapies can be developed, which could take several months. "There have been tests that show when a person is injected with the antibodies, that then stimulates and promotes their immune system against that disease," Cuomo said. "It's only a trial. It's a trial for people who are in serious condition, but the New York State Department of Health has been working on this with some of New York's best health care agencies, and we think it shows promise, and we're going to be starting that this week." New York has seen a dramatic rise in coronavirus cases in recent days, with about 20,000 confirmed cases and more than 150 deaths as of Monday afternoon, more than in any other state. State health officials said they expect to get approval from the Food and Drug Administration to begin the plasma trial in the coming days. A spokesman for the FDA confirmed to NBC News that the agency is "working expeditiously to facilitate the development and availability" of convalescent plasma. And at a White House briefing last week, FDA Commissioner Stephen Hahn singled out the treatment as a promising option to fight the disease in the near term.

|
Scooped by
Juan Lama
|
China’s state-owned medical products maker is collecting plasma from the blood of people who have recovered from the novel coronavirus after discovering it helped critically ill patients. China National Biotec Group Co. has been using this plasma, which contains highly potent antibodies, to treat some seriously ill patients since Feb. 8, the company said in an statement on its official WeChat account Thursday night. It claimed that those receiving the treatment improved within 24 hours, with reduced inflammation and viral loads along with better oxygen levels in the blood. Drugmakers and the Chinese authorities are racing to develop a cure for the contagion that has so far killed more than 1,300 people, infected over 63,000 and has no approved treatment or vaccine. China has also unleashed a flurry of clinical trials — as many as 77 have been registered — in a bid to curb the outbreak that’s threatening its economic growth. The announcement triggered a rally in the stocks of blood product makers on Friday. Beijing Tiantan Biological Products Corp., Shanghai RAAS Blood Products Co. and Hualan Biological Engineering Inc. jumped to their daily trading limit of 10%, according to data compiled by Bloomberg. “Patients who have recovered from the novel coronavirus pneumonia, will generate antibodies that can kill and remove the virus,” the company said in the statement, citing unidentified experts. “In the absence of a vaccine and specifically targeted drugs, using this plasma is the most effective way to treat the infection and can significantly reduce the death toll.” China National Biotech also called on people who have recovered from the disease to donate their plasma in a separate statement. China’s National Health Commission has listed plasma among treatment measures for critically ill patients in its latest treatment guideline....
|


 Your new post is loading...
Your new post is loading...