Background
Short- and long-term implications of SARS-CoV-2 on the quality of the sperm and the results of this on fertility remain largely unknown due to lack of longitudinal studies. In this longitudinal observational cohort study, we aimed to analyse the differential effect and the impact of SARS-CoV-2 infection on different semen quality parameters.
Methods
Sperm quality was assessed using the World Health Organization criteria, DNA damage to sperm cells by quantifying the DNA fragmentation index (DFI) and the high-density stainability (HDS), IgA- and IgG-anti-sperm antibodies (ASA) were assessed with light microscopy.
Findings
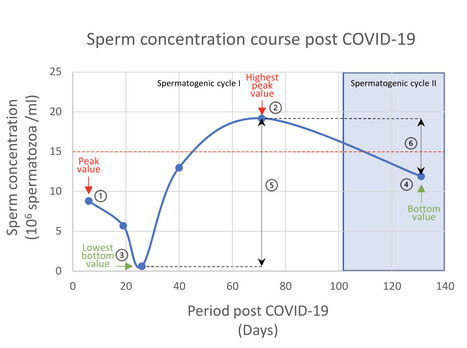
SARS-CoV-2 infection was associated with sperm parameters that were independent of spermatogenic cycle like progressive motility, morphology, DFI and HDS, as well as spermatogenic cycle dependent parameters such as sperm concentration. Detection of IgA- and IgG-ASA allowed classification of patients in three different groups according to its sequence of appearance in sperm during post-COVID-19 follow-up. The maximum progressive motility was lowest during follow-up in patients without ASA (41.9%), intermediate in patients with only IgA-ASA (46.2%) and highest inpatients who had both IgA- and IgG-ASA (54.9%).
Interpretation
SARS-CoV-2 infection was associated with changes of all analysed sperm parameters to a different degree which is also observed in their return to normality and is suggestive of individual variations in the patient's immune system performance. Firstly, sperm production is decreased through temporal immune mediated arrest of active meiosis, and secondly immune induced sperm DNA damage prevents fertilization if transferred to the oocyte. Both mechanisms are temporal, and most sperm parameters return to baseline after infection.
Evidence before this study
Although some studies have shown the presence of SARS-CoV-2 virus in the testis of deceased COVID-19 patients and during the acute phase of the infection, many studies reported the absence of the virus in semen shortly after COVID-19. Despite the potential risk for sexual transmission of the SARS-CoV-2 virus therefore being low, short- and long-term effects on the quality of the sperm and the impact of this on fertility remain largely unknown due to lack of longitudinal studies.
Added value of this study
This large follow-up study of post COVID-19 patients demonstrated a harmful effect of SARS-CoV-2 infection on different sperm parameters through immune response. Recovery of the different sperm parameters varied between patients, could take more than one-year post infection, and depended on the initial immune response of the patient. Our data show for the first time that peak progressive sperm motility post-COVID-19 is dependent on the patient's antibody response. Patients producing both IgA/IgG-ASA had the fastest and most complete recovery, while patients without IgA/IgG-ASA experienced slow and incomplete recovery. Conversely, harmful sIgG-N in blood only significantly correlated with higher sperm DNA damage (DFI). Our longitudinal data show that the reduction in sperm concentration after SARS-CoV-2 infection is not caused by fever but rather by the immunological response that follows afterwards. The main goal of the immune response after a SARS-CoV-2 infection is to protect cell populations amplified through meiosis. Firstly, sperm production is decreased through temporal arrest of active meiosis, and secondly the sperm DNA is damaged preventing fertilization if transferred to the oocyte. Both mechanisms are temporal, and most sperm parameters return to baseline after infection.
Implications of all the available evidence
Detection of IgA and IgG ASA that bind the tail of spermatozoa allowed a novel classification of patients in 3 different groups according to the detection and sequence of appearance in sperm during post COVID-19 follow-up. Detection of harmful (sIgG-N) and protective (RBD-IgG) antibodies in both serum and sperm (IgA/IgG-ASA) as well as the measurement of sperm DNA damage in sperm should be promoted in fertility assessment. While we showed that the humoral immunity plays a key role in modulating sperm parameters after SARS-CoV-2 infection, future research will focus on how the cellular immunity and t-cell mediated response can also impact fertility.
Published in eBioMedicine (July 2023):


 Your new post is loading...
Your new post is loading...