Research and publish the best content.
Get Started for FREE
Sign up with Facebook Sign up with X
I don't have a Facebook or a X account
Already have an account: Login
Latest advances in immunopathology diagnosis and treatment
Curated by
Alfredo Corell
 Your new post is loading... Your new post is loading...
 Your new post is loading... Your new post is loading...
|
|



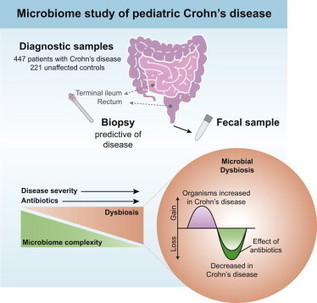








Inflammatory bowel diseases (IBDs), including Crohn’s disease (CD), are genetically linked to host pathways that implicate an underlying role for aberrant immune responses to intestinal microbiota. However, patterns of gut microbiome dysbiosis in IBD patients are inconsistent among published studies. Using samples from multiple gastrointestinal locations collected prior to treatment in new-onset cases, we studied the microbiome in the largest pediatric CD cohort to date. An axis defined by an increased abundance in bacteria which include Enterobacteriaceae, Pasteurellacaea, Veillonellaceae, and Fusobacteriaceae, and decreased abundance in Erysipelotrichales, Bacteroidales, and Clostridiales, correlates strongly with disease status. Microbiome comparison between CD patients with and without antibiotic exposure indicates that antibiotic use amplifies the microbial dysbiosis associated with CD. Comparing the microbial signatures between the ileum, the rectum, and fecal samples indicates that at this early stage of disease, assessing the rectal mucosal-associated microbiome offers unique potential for convenient and early diagnosis of CD.
Divulgative news: http://www.sys-con.com/node/3009818