Elimination of Germinal Center-Derived Self-Reactive B Cells Is Governed by the Location and Concentration of Self-Antigen
Immunity, 08 November 2012
10.1016/j.immuni.2012.07.017
Authors
Tyani D. Chan, Katherine Wood, Jana R. Hermes, Danyal Butt, Christopher J. Jolly, Antony Basten, Robert Brink
Summary
Secondary diversification of the B cell repertoire by immunoglobulin gene somatic hypermutation in the germinal center (GC) is essential for providing the high-affinity antibody specificities required for long-term humoral immunity. While the risk to self-tolerance posed by inadvertent generation of self-reactive GC B cells has long been recognized, it has not previously been possible to identify such cells and study their fate. In the current study, self-reactive B cells generated de novo in the GC failed to survive when their target self-antigen was either expressed ubiquitously or specifically in cells proximal to the GC microenvironment. By contrast, GC B cells that recognized rare or tissue-specific self-antigens were not eliminated, and could instead undergo positive selection by cross-reactive foreign antigen and produce plasma cells secreting high-affinity autoantibodies. These findings demonstrate the incomplete nature of GC self-tolerance and may explain the frequent association of cross-reactive, organ-specific autoantibodies with postinfectious autoimmune disease.
A summary in Garvan Institute:
http://www.garvan.org.au/news-events/news/how-infection-can-trigger-autoimmune-disease.html
A previous publication of this theory:
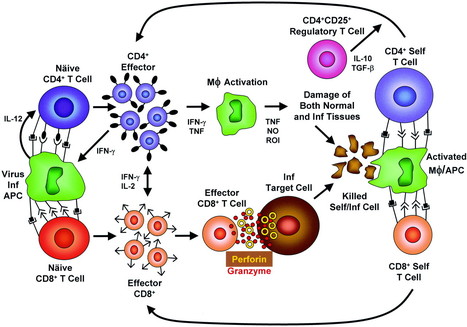
Mechanisms for the induction of autoimmunity by infectious agents
Kai W. Wucherpfennig
Published in Volume 108, Issue 8 (October 15, 2001)
J Clin Invest. 2001;108(8):1097–1104. doi:10.1172/JCI14235.
http://www.jci.org/articles/view/14235
 Your new post is loading...
Your new post is loading...
 Your new post is loading...
Your new post is loading...