Researchers have found a method to repair the gene mutation causing agammaglobulinemia, an immunodeficiency disease that almost exclusively affects boys and in which the body lacks the ability to produce immunoglobulins (gamma globulin). The disease is characterized by recurring bacterial infections, mainly in the respiratory system, and persons who suffer from the illness currently need life-long gamma globulin treatment.
Research and publish the best content.
Get Started for FREE
Sign up with Facebook Sign up with X
I don't have a Facebook or a X account
Already have an account: Login
Latest advances in immunopathology diagnosis and treatment
Curated by
Alfredo Corell
 Your new post is loading... Your new post is loading...
 Your new post is loading... Your new post is loading...
|

Gilbert C FAURE's curator insight,
October 2, 2013 3:18 PM
will help medical students understand the RAG is a useful enzyme |






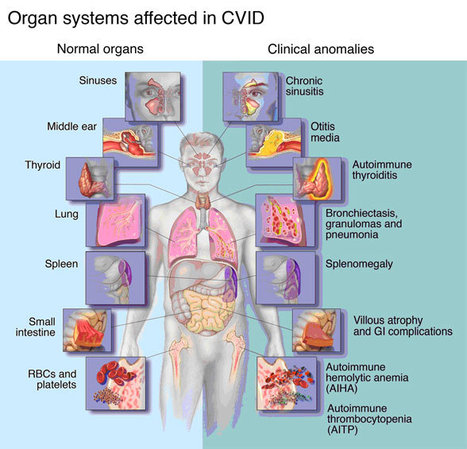














X-linked agammaglobulinemia (XLA) is an inherited immunodeficiency that results from mutations within the gene encoding Bruton’s tyrosine kinase (BTK). Many XLA-associated mutations affect splicing of BTK pre-mRNA and severely impair B cell development. Here, we assessed the potential of antisense, splice-correcting oligonucleotides (SCOs) targeting mutated BTK transcripts for treating XLA. Both the SCO structural design and chemical properties were optimized using 2′-O-methyl, locked nucleic acid, or phosphorodiamidate morpholino backbones. In order to have access to an animal model of XLA, we engineered a transgenic mouse that harbors a BAC with an authentic, mutated, splice-defective human BTK gene. BTK transgenic mice were bred onto a Btk knockout background to avoid interference of the orthologous mouse protein. Using this model, we determined that BTK-specific SCOs are able to correct aberrantly spliced BTK in B lymphocytes, including pro–B cells. Correction of BTK mRNA restored expression of functional protein, as shown both by enhanced lymphocyte survival and reestablished BTK activation upon B cell receptor stimulation. Furthermore, SCO treatment corrected splicing and restored BTK expression in primary cells from patients with XLA. Together, our data demonstrate that SCOs can restore BTK function and that BTK-targeting SCOs have potential as personalized medicine in patients with XLA.
GO TO THE JOURNAL OF CLINICAL INVESTIGATION MANUSCRIPT:
http://www.jci.org/articles/view/76175